Edit Graphs
The sequence Edit Graph is very useful for understanding what came off
the sequencer - although you may need to play with the thresholds to find a
sweet spot for hiding the noise. Using run.sh calls the pipeline with a
minimum abundance 10, which would give large noisy edit graphs. Instead, we
build them using a minimum abundance of 100, giving files
SPH16S.edit-graph.a100.xgmml and MOL16S.edit-graph.a100.xgmml, and
additional graphs for the mock community samples alone.
My main conclusion from the figures below is that the THAPBI PICT default
onebp classifier is reasonable for these mock communities markers.
However, the MOL16S database needs considerable expansion for use on the
environmental samples. Perhaps updating this example in 5 years time there
will be enough published markers to assign species to all the unknowns here?
SPH16S
First SPH16S, where there are just the three samples for the mock communities. Each is expected to have three species Sphaerium simile, Sphaerium corneum and Pisidium compressum only. With a minimum abundance threshold of 100, we get three nice clear graph components, and a few single nodes:

Next, using all the samples but again a sample level minimum abundance 100:
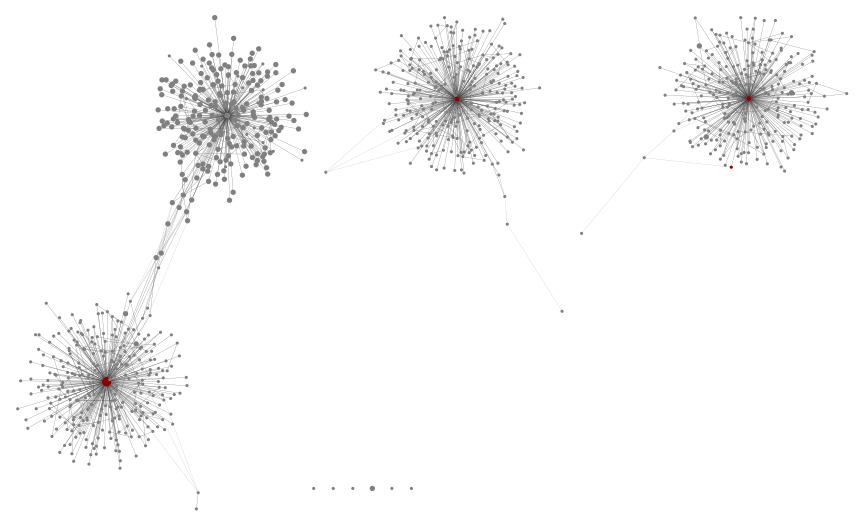
Very little change except the addition of a fourth cluster, some base pairs away from the Sphaerium simile component and centred on this sequence:
>79b63a2ef96b839ae3263369f8d390b9
ACGTGGAAAAAACTGTCTCTTTTGTATAAAAAGAAGTTTATTTTTAAGTGAAAAAGCTTAAATGTTTATAAAAGACGAGA
AGACCCTATCGAACTTAAATTATTTGTTTAAATTTTTAAATAAAAAAAAGTTTAGTTGGGGAAACTTAAAGTAAAAAGTA
ACGCTTTATTTTTTTGTCAGGAGCCTGTAGTATGGAAAAATGAAAAAGTTACCGTAGGGATAACAGCGCTTTCTTCTCTG
AGAGGACTAATTAAAGAGTT
This is likely another Sphaerium species, NCBI BLAST suggests Sphaerium
striatinum - with AF152045.1 just two base pair away. This is in our
reference database file SPH16S.fasta:
>AF152045.1 Sphaerium striatinum
ACGTGGAAAAAACTGTCTCTTTTGTATAAAAAGAAGTTTATTTTTAAGTGAAAAAGCTTAAATGTTTATAAAAGACGAGA
AGACCCTATCGAACTTAAATTATTTGTTTAAATTTTTAAATAAAAAAAAGTTTAGTTGGGGAAACTTAAAGTAAAAATTA
ACGCTTTATTTTTTTGTCAGGAGCCTGTACTATGGAAAAATGAAAAAGTTACCGTAGGGATAACAGCGCTTTCTTCTCTG
AGAGGACTAATTAAAGAGTT
MOL16S
For MOL16S, starting with an edit graph of just the seven MOL16S samples, and a minimum abundance threshold of 100, we see:
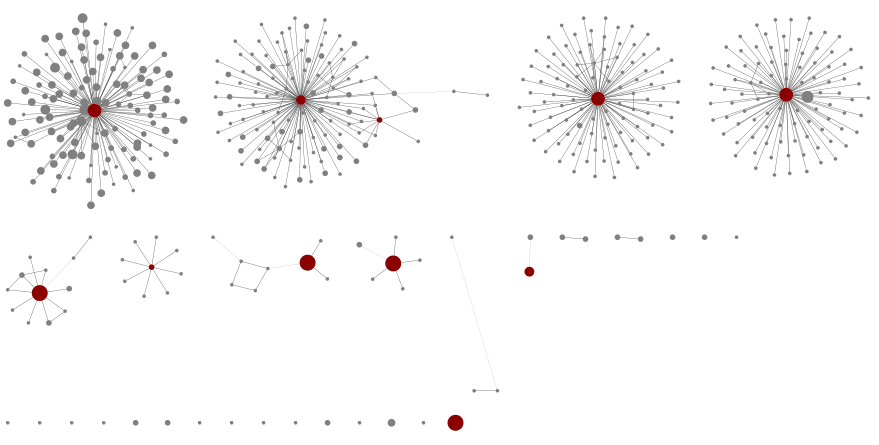
Four large components representing species with lots of variants, with red central nodes in our database. Other less diverse graph components for the remaining species, and a selection of isolated unknowns.
Next, using all the samples but again a sample level minimum abundance 100:
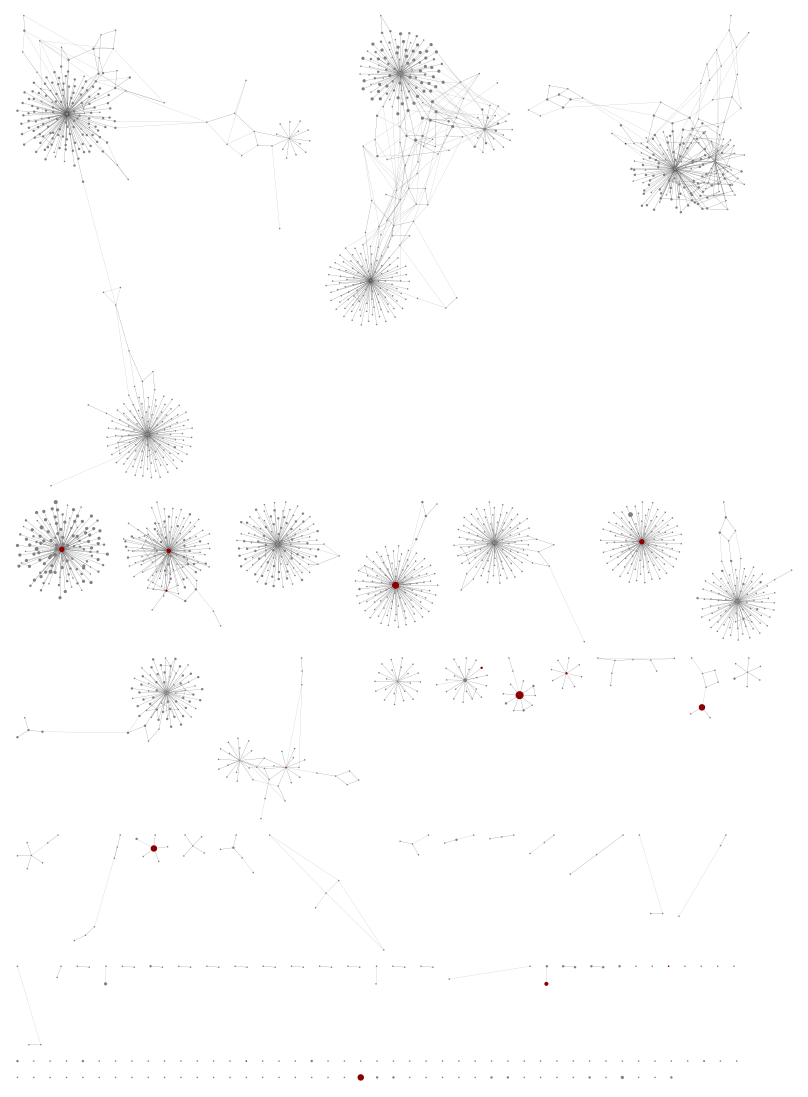
Suddenly we see dozens of new components, most of which have no references (coloured nodes) representing likely unknown species.
Conclusion
I will close by quoting the end of Klymus et al. (2017):
The present study further demonstrates that metabarcoding data are only as good as the sequence and taxonomic information provided on genetic databases. Increased collaboration among taxonomists and molecular systematists is required in order to gain maximum benefits of this developing tool.
I agree - these markers seem to work, but there are still too many unknown sequences.